
New routine enables access to 1.5 million Swedish COVID-19 samples for research
Thanks to a unique collaboration with the Swedish regions and the National Pandemic Center (NPC) at Karolinska Institutet, Biobank Sweden is launching a national COVID-19 sample collection for research. With a new national application process, the purpose is to foster research related to COVID-19 through easier access to samples stored at the NPC.
Many samples from the Swedish regions are sequenced at the National Pandemic Center (NPC) at Karolinska Institutet. NPC also store samples based on the Swedish Infection Protection Act, which over time has resulted in a “national” COVID-19 sample collection. As the sample collection offers a valuable resource for research, Biobank Sweden together with the Swedish regions and NPC, has developed a new national routine for researchers to apply for and get access to the samples stored at NPC. In total, the sample collection at the NPC contains more than 1.5 million COVID-19 samples from all over Sweden, covering the whole pandemic time period.
– This sample collection offers a unique opportunity for retrospective analysis of disease transmission and spread, and related research questions, for which there were no possibility or resources to analyze under the most intensive phases of the pandemic, says Jessica Alm, Associate Professor, Head of Unit at National Pandemic Center, Karolinska Institutet.
New national application process
To make the sample collection at NPC accessible to research, a new national application process is now in place, where all regions are collaborating to enable a common application process regardless of where samples are stored (Figure 1). A new application form has been created as part of the new process, specific for the COVID-19 collection at NPC.
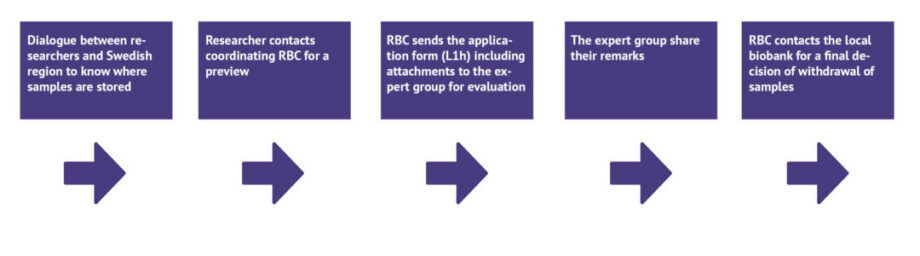
As part of this national streamlined process, Biobank Sweden has appointed two Regional Biobank Centres (RBC) to manage and coordinate the application process; RBC Syd and RBC Mellansverige. With this set-up, the aim is to have a more efficient handling of applications, where the coordinating RBCs are responsible for the overall managing and regulatory review of submitted applications in accordance with the Swedish Biobank Act. In addition, a new expert review board has been appointed responsible for the scientific evaluation of applications for samples from the collection at NPC. Each region still retains the legal responsibility of its own samples and consequently has the final decision regarding access to and withdrawal of samples from the collection at NPC.
– The centralized storage of COVID-19 samples at NPC makes it easier for researchers to access samples and improves the sample quality, as samples from several regions are handled and stored in a standardized manner, and selection for research is conducted from one single location. It will also be easier and more efficient to handle applications as they are managed through a centralized process, says Katarina Järlestedt, Biobank coordinator at Region Dalarna.
COVID-19 portal will visualize the sample collection
In collaboration with SciLifeLab, the sample collection at NPC is published digitally within the sample collection register at the Swedish COVID-19 and Pandemic Preparedness Portal. This will provide a natural point of access for researchers and give an overview of the sample collection along with concrete information on the process and routine for accessing the sample collection. The data portal is an established platform for visualizing other COVID-19-related sample collections in Sweden, hence, the digital infrastructure is already in place. The goal is to stimulate and contribute to increased COVID-19 research, also beyond the NPC hosted sample collection.
– We hope that this extraordinary sample collection will be used by researchers within the field to gain new knowledge, not only for COVID-19, but also useful insights of value for future pandemics. Ideally this can make us better prepared, and hopefully reduce the disease burden and costs for society, says Lars Engstrand, Professor/senior consultant, Director at National Pandemic Center, Karolinska Institutet.
All applications for access to samples stored at NPC require an approval from the Swedish Ethical Review Authority (Etikprövningsmyndigheten), and research must be COVID-19-related.
How to apply for samples stored at NPC
Application for samples stored at NPC can be done by using documents L1 and L1H. The documents are only available in Swedish.
För more information, see the following documents:

About National Pandemic Center (NPC) at Karolinska Institutet
National Pandemic Center (NPC) at Karolinska Institutet was founded in March 2020 as a COVID-19 diagnostic laboratory at the Karolinska Institutet Centre for Translational Microbiome Research (CTMR). The aim was to support Swedish healthcare with large scale analysis of COVID-19 by nucleic acid detection of SARS-CoV-2, i.e., PCR-testing, in a critical situation where testing capacity in Sweden did not match the increased demands caused by the pandemic.
Since 2021, large scale whole genome sequencing (WGS) of SARS-CoV-2 constitutes the main activities at NPC, contributing to the national surveillance of the virus evolution under agreement with the Swedish Public Health Agency (PHA)(Folkhälsomyndigheten). Due to this task, NPC has received COVID-19 samples from all over Sweden, and thereby enabled this centralized sample collection.
During 2022, NPC was also appointed as a national COVID-19 diagnostic preparedness laboratory by Folkhälsomyndigheten. Since the start of the pandemic, NPC has conducted over 600 000 diagnostic PCR-tests; over 130 000 WGS; and now has over 1.5 million samples stored. Through these tasks, NPC has established well-functioning collaborations and logistic chains with the Swedish regions, and productive cooperations with different stakeholders, such as Biobank Sweden.



